Here ate today’s CHM 110 notes. These notes discuss the predictions of Dalton’s theory, subatomic particles, the nuclear model of the atom, and the periodic table.
Monthly Archives: January 2009
CHM 111: Board notes for 1/28/09
Here are yesterday’s CHM 111 notes. These notes discuss the classification of solids by structure and the units for calculating the concentration of a solution.
CHM 100: Board notes for 1/28/09
Here are today’s CHM 100 class notes. These notes cover how to deal with the number zero in measurements and how to round answers when adding measured numbers.
CHM 110: Board notes for 1/27/09
Here are the notes from today’s CHM 110 class. These notes cover math with significant figures, exact numbers, and an introduction to Dalton’s atomic theory.
CHM 111: Board notes from 1/26/09
Here are today’s class notes. These notes cover intermolecular forces in liquids and how they affect liquid properties. They also cover the kinds of forces present in the solid state.
CHM 100: Board notes for 1/26/09
Here are today’s CHM 100 notes. These notes cover a few more dimensional analysis examples, then introduce accuracy, precision, and the concept of significant figures.
CHM 110: Board notes for 1/22/09
Here are the notes from Thursday’s CHM 110 class. These notes introduce the concepts of accuracy, precision, and significant figures.
CHM 111: Board notes from 1/21/09
Here are the notes from Wednesday’s CHM 111 class. These notes continue our discussion of valence bond theory from Chapter 10, then introduce the phases of matter from Chapter 11.
We discusses several chlorine-containing hydrocarbons during our talk about how double bonds work in valence bond theory. Here they are.
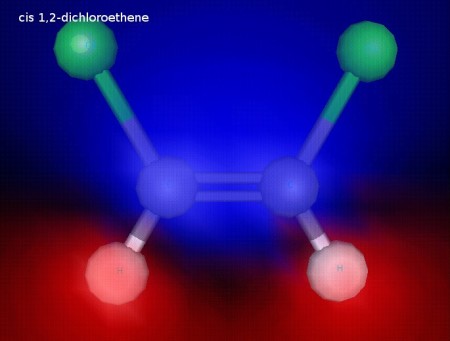
This is cis 1,2-dichloroethene. Both of the chlorine atoms are on the same side of the C=C bond. The chlorine atoms are fixed in this position because the double bond does not allow free rotation, since rotation would break the pi component of the double bond. This illustration shows electrostatic potential, and we can see that this molecule is polar.
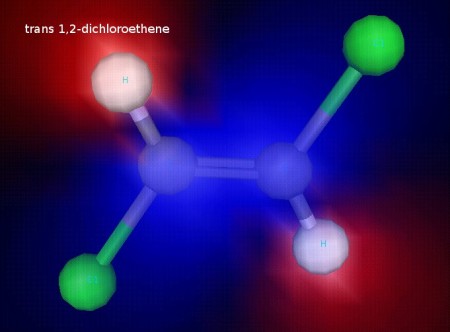
This is trans 1,2-dichloroethene. The chlorine atoms are on opposite sides of the C=C bond. The chlorine atoms are fixed in this position because the double bond does not allow free rotation, since rotation would break the pi component of the double bond. This illustration shows electrostatic potential, and we can see that this molecule is nonpolar. It also has observably different physical properties (like boiling point) from the cis form.
CHM 100: Board notes from 1/21/09
Here are the notes from today;s CHM 100 class. These notes discuss how to convert between units with dimensional analysis.
MLK Holiday
Monday 1/19/09 is a holiday. The school will be closed, and no classes or labs will meet. Here’s how the holiday will affect your chemistry schedule.
CHM 100 students
- No lecture on Monday 1/19
- Section 1 lab will not meet on 1/19. You will perform the introductory experiment on 1/26.
- Section 2 lab will meet on Tuesday 1/20. You will perform the introductory experiment. I will hand out a copy of the experiment when you get to lab, but if you want to look it over before coming in, here it is: [CHM 100 Introductory Experiment]
CHM 110 students
- Your class schedule is unaffected by the holiday. You will perform the introductory experiment (the same one as the CHM 100 class) on Thursday 1/22.
CHM 111 students
- No lecture on Monday 1/19
- You will perform Experiment 9B on Wednesday 1/21. Remember that the prelab for the experiment (pages 177-178 inyour lab manual) is due at the beginning of the lab period. Please have it ready when you walk in. Also, don’t forget your lab manual and notebook on Wednesday.