Here is a gallery of examples of the various molecular shapes rendered in 3D with a molecular modeling program.. Some of these renderings are in your notes already, but some were not.
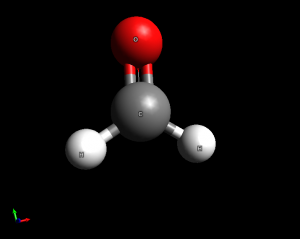
- Formaldehyde (CH2O). This is a trigonal planar molecule. Bond angles are 120 degrees.
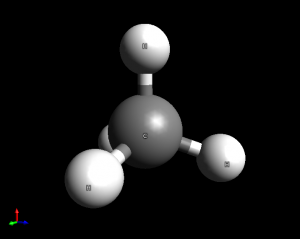
- Methane (CH4). This is a tetrahedral molecule with 109.5 degree bond angles.
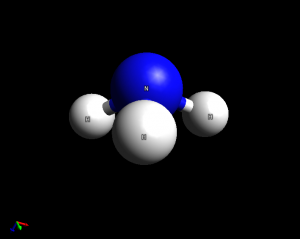
- Ammonia (NH3). This is a pyramidal molecule. Bond angles are similar to the tetrahedral 109.5 degrees due to the presence of a lone pair of electrons on the nitrogen atom.
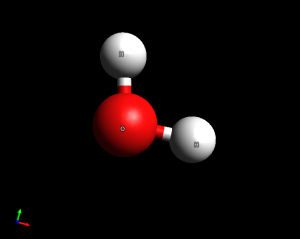
- Water (H2O). This is a bent molecule. Bond angles are similar to the tetrahedral 109.5 degrees, since there are two lone pairs on the oxygen atom that act like other atoms.
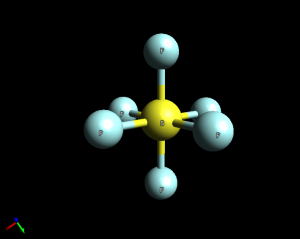
- Sulfur hexafluoride (SF6). This is an octahedral molecule with 90 degree bond angles. Note: This geometry can only be assumed by an expanded valence molecule.