Monthly Archives: September 2008
CHM 100: Board notes for 9/29/08
CHM 110: Board notes for 9/24/08 and 9/26/08
Here are the notes from our CHM 110 classes on Wednesday and today. These notes cover chemical calculations: basic stoichiometry, limiting reactant, and percent yield.
[CHM 110: 9/24/08 and 9/26/08 notes]
Any questions? Ask here!
CHM 100: Board notes for 9/24/08 and 9/26/08
Here are the notes for CHM 100 from Wednesday abd today. These notes cover Daton’s atomic theory, atomic terms, and an introduction to the periodic table.
[CHM 100: 9/24/08 and 9/26/08 notes]
Any questions? Ask here!
New student club
The missing-text blues
A few people have told me that when they try to print the board notes, they’re having trouble with fonts disappearing. I’ve reproduced the problem myself in one of the student conputer labs in the 5000 muilding. I have discovered a workaround for the problem – use a different PDF viewer. Here’s one that I tested in the school computer labs: Sumatra PDF
If you’d like to use Sumatra PDF in one of the school labs, then download the Sumatra ZIP file to your desktop, and double-click it. Copy the single file inside the folder that opens up to the desktop, then double-click it to run it. You can also copy that file to a USB pen drive and take it with you to any other computer.
To view or print the notes for your class, just save them to your Desktop (right-click on the link), then open them with the Sumatra PDF viewer.
Any questions? Ask here!
Melamine in the news
It looks like there’s trouble in China. Supplies of milk powder meant for Chinese infants are tainted with melamine – the same chemical found in last year’s dog-food scare.
On Sept. 17, China’s minister of health, Chen Zhu, announced that three babies had died, more than 150 were suffering from acute kidney failure, and an additional 6,000 infants had become sick after drinking milk made from milk powder tainted with melamine.
Now you might be wondering why on earth a company would add melamine to powdered milk. Could it have been accidental? Or was there some reason to add this compound?
Consider this: The amount of protein in foods is often determined based on total nitrogen content. That’s because proteins contain a fairly regular amount of nitrogen – about 16% by mass. Take the total nitrogen content, multiply by a conversion factor, and you have a good estimate of protein content of the food.
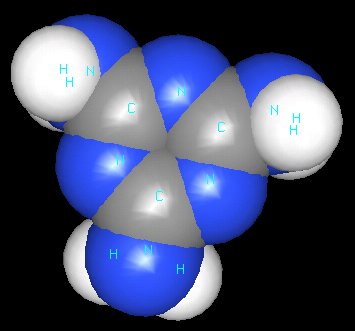
Take a look at melamine.
Simple assay methods for total nitrogen content can’t distinguish the nitrogen in melamine – which is about 67% nitrogen by mass – from the nitrogen in proteins.
The tested substance appears to have a higher protein content than it actually does – since analysts assume that almost all the measured nitrogen comes from actual protein. This is a reasonable assumption for uncontaminated materials, but is open to abuse by the amoral.
CHM 100: Board notes for 9/22/08
CHM 100: Board notes for 9/19/08
Software update
This morning, I updated the WordPress software that powers this site. If the update went well, you won’t notice any differences in using the site (it was a minor bug-fix update).
If you notice any problems with the site like broken links or buttons not doing what they’re supposed to, please let me know.