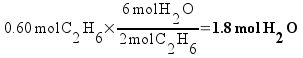
Introduction
We've discussed how to solve basic stoichiometry problems - those where you're given the amount of only one substance. However, in some situations, you might be given a problem to solve where the amount of more than one substance is specified. In that case, how do you determine when the chemical reaction will end?
The limiting reactant
A chemical reaction will end when any reactant is completely consumed – no matter whether other reactants are left or not. Consider the combustion of a magnesium strip.
2 Mg(s) + O 2 (g) --> 2MgO (s)
If you burn a small magnesium strip using the oxygen in room air, you would run out of magnesium long before you ran out of oxygen. Otherwise, you'd kill yourself from lack of oxygen during this or any other combustion reaction! Magnesium is the limiting reactant in the experiment because it is used up completely. Oxygen is in excess because there is oxygen left after the reaction stops.
(You can die if you are present in a room where combustion in the open air is occurring and oxygen is the limiting reactant!)
The chemical reaction stops when we run out of the limiting reactant. No more product is produced. Therefore, the amount of product produced in the reaction is controlled solely by the limiting reactant.
Application and problem solving
You could run into a limiting reactant situation anywhere - even outside the lab. The most common place to run into this is, of course, in the kitchen (as in the book's example). Inside the lab, if you're trying to make as much of a product as possible and have a fixed amount of each reactant, you'll have to solve a limiting reactant problem.
Basically, anytime you've been given the amount of more than one reactant in a stoichiometry problem, you have a limiting reactant problem.
For an example, we'll use a simple problem based on a reaction we've worked with previously.
How many moles of water are produced if 0.60 mol ethane (C2H6) is reacted with 1.5 moles of molecular oxygen (O2)? The reaction is:
2 C2H6 + 7 O2 --> 4 CO2 + 6 H2O
This problem is simplified in that moles (instead of mass) are provided and asked for, but it illustrates the concept of a limiting reactant.
Normally, we'd find formula weights and convert all given masses to moles , but this problem does not require it.
What we have to do now is this - determine the limiting reactant. What runs out first, the 0.60 mol ethane or the 1.5 mol oxygen? Be careful to avoid the common mistake of observing that there are more moles of oxygen than ethane and reaching the conclusion that ethane must to be the limiting reactant because there's less of it! This reaction consumes more oxygen than ethane on a molar basis. Look at the coefficients of oxygen and ethane!
So how do we determine which one runs out first? The simplest way is to actually calculate how many moles of product that could be theoretically formed from each reactant . That is, calculate how much water could be formed from the oxygen if there was enough ethane and how much water could be formed fro m the ethane if there was enough oxygen.
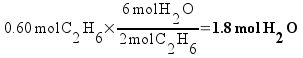
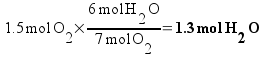
We can make less product from the oxygen. All the oxygen is gone when we've formed 1.3 moles of water, so the reaction stops there. Oxygen is the limiting reactant, and 1.3 moles of water are formed in the reaction!
Summary
In this note pack, we have illustrated using a simple example the concept of a limiting reactant. You should know that a limiting reactant is the one that runs out first in a chemical reaction. Once this reactant is gone, no more product can be produced because the reaction stops.. To solve a limiting reactant problem, you calculate the amount of product that can be produced from all the reactants given. The reactant that produces the smallest amount of product is the limiting reactant.
All original site content ©2007 Charles Taylor. Page updated: November 28, 2007.